
On June 6, 2025, the international top-tier journal Gut (3-year average Impact Factor: 26.4; CAS Q1 Top Journal) published a groundbreaking study titled “NLRP6 deficiency enhances macrophage-mediated phagocytosis via E-Syt1 to inhibit hepatocellular carcinoma progression.”
This collaborative research was led by Professor Lu Yinying from the Peking University 302 Clinical Medical School (The Fifth Medical Center of PLA General Hospital) and Professor Shi Yuanyuan from Shenzhen Cell Valley. The study unveils, for the first time, the molecular mechanism by which the innate immune receptor NLRP6 regulates hepatocellular carcinoma (HCC) progression by modulating macrophage phagocytic function. This discovery identifies a novel target for developing macrophage-based immunotherapies for liver cancer.
Authorship & Collaborations:
First Author: Dr. Li Shuang (Peking University Health Science Center)
Second Author: Dr. Fu Yuchen (Shenzhen Cell Valley)
Collaborating Institutions: Beijing Gaobo Hospital, Hadassah Hebrew University Medical Center (Israel), Tsinghua University Synthetic Biology Center, among others.
Liver cancer remains a leading cause of cancer-related deaths globally, with current targeted therapies and immune checkpoint inhibitors often limited by drug resistance.
Clinical Correlation: Patients with low NLRP6 expression in macrophages demonstrated significantly prolonged survival rates.
In Vivo Efficacy: In mouse tumor models, a single infusion of Nlrp6⁻/⁻ macrophages inhibited tumor growth by 70%, with the anti-tumor effect proven to be dependent on the E-Syt1 signaling pathway.
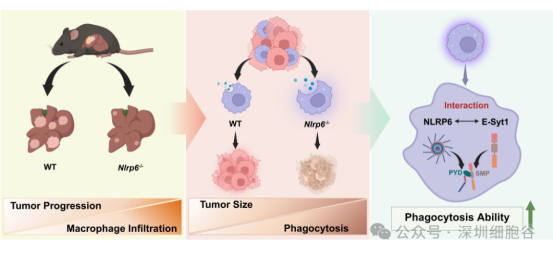
The study reveals a previously unknown negative feedback loop:
Molecular Interaction: NLRP6 binds directly to the key endocytic protein E-Syt1 via its PYD domain interacting with E-Syt1’s SMP domain.
Functional Outcome: Disruption of this axis (via NLRP6 deficiency) significantly enhances macrophage phagocytosis of tumor cells.
In Vitro Validation: NLRP6-deficient macrophages exhibited a 2.5-fold increase in phagocytic activity against HCC cells compared to wild-type controls.
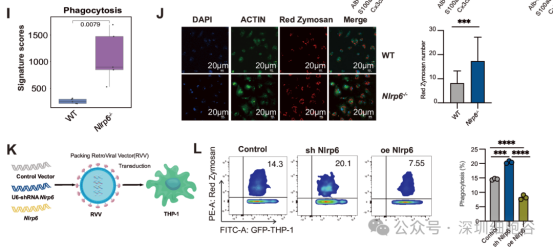
Leveraging Shenzhen Cell Valley’s proprietary retroviral vector platform, the team successfully engineered NLRP6-edited macrophage lines with superior anti-tumor capabilities.
This work not only clarifies NLRP6’s critical role in regulating macrophage anti-tumor function but also charts a new course for the clinical application of CAR-M (Chimeric Antigen Receptor Macrophage) therapies in treating liver cancer.
The PLA General Hospital and Shenzhen Cell Valley have jointly filed international patents covering these original therapeutic targets centered on NLRP6 and E-Syt1.
We warmly invite forward-thinking biopharmaceutical enterprises from China and abroad to collaborate with us. Together, we can accelerate the joint development and global translation of these next-generation cell therapies, bringing hope to patients worldwide.
This landmark publication underscores the power of strategic academia-industry partnerships in driving precision medicine forward.

 Scan to follow our latest news
Scan to follow our latest news Working hours: Monday to Friday, 9:00-18:00
Contact:Ms. Lai
Email:laijiaqi@sz-cell.com
Address:No. 1, Rongtian Road, Jinsha Community, Kengzi Street, Pingshan District, Shenzhen, China (Hepure Biomedical Ecological Park)