On May 26, 2025, the research team at Shenzhen Cell Valley published a study titled “Construction of stable packaging cell lines for large-scale industrial BaEV-enveloped retroviral vector production” in the journal Frontiers in Immunology (CAS Zone 2, Impact Factor 5.7).
The paper details the construction of a stable packaging cell line for BaEV-enveloped retroviral vectors. The retroviral vectors produced by this cell line are particularly well-suited for transducing primary cells such as NK cells and T cells. Notably, the BaEV stable packaging cell line constructed by the company was deposited with the Guangdong Provincial Center for Microbial Culture Collection on July 17, 2024, and relevant patents have already been granted.
Shenzhen Cell Valley is committed to providing GMP-grade retroviral vectors and cell therapy products that are highly safe, high-quality, and cost-effective, ensuring the safety and efficacy of cell therapy products and driving the overall development of the cell and gene therapy industry.

1. Construction of the HEK293T-gag-pol-ASCT-1&2-KO Cell Line
The team first utilized plasmid transfection to introduce the structural protein gag-pol and the BaEV envelope protein required for retroviral production into HEK293T cells. These cells were engineered to stably express these proteins while simultaneously knocking out the ASCT-1/2 receptors. This process yielded the HEK293T-gag-pol-ASCT-1&2-KO cell line.
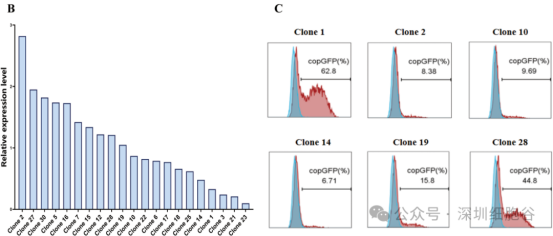
(See Figure 2 & 3: Construction of HEK293T-gag-pol and HEK293T-gag-pol-ASCT-1&2-KO cell lines)
2. Development of the BaEV Stable Packaging Cell Line (BaEV-PackRV)
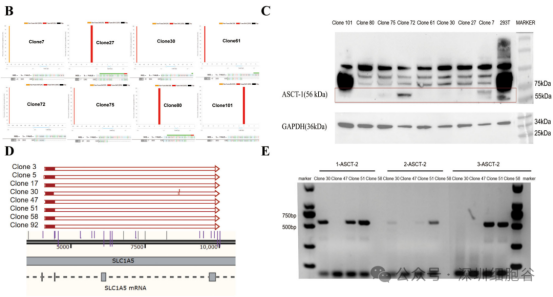
Building on the previous step, the team transfected the BaEV envelope plasmid into the knockout cell line. Through antibiotic resistance screening, single-cell clones were isolated. These clones underwent transient transfection titer testing to identify those with high viral titers and unaffected growth status. The selected clone was designated as the BaEV retroviral vector stable packaging cell line (BaEV-PackRV).
(See Figure 4: Construction of the HEK293T-gag-pol-ASCT-1&2-KO-BaEV cell line)
3. Superior Transduction Efficiency Compared to GaLV
Using this stable packaging cell line, the team constructed both BaEV-CD19 CAR-RV and traditional GaLV-CD19 CAR-RV vectors. These were used to infect primary T cells and NK cells.
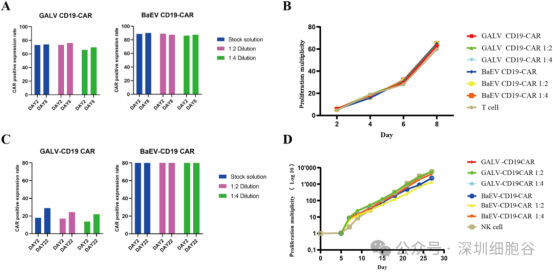
· Results: Compared to GaLV-CD19 CAR-RV, the BaEV-CD19 CAR-RV demonstrated excellent transduction efficiency in primary cells even at extremely low titers and low Multiplicity of Infection (MOI).
(See Figure 5: Comparison of BaEV/GaLV CD19-CAR RV transduction in human primary T cells)
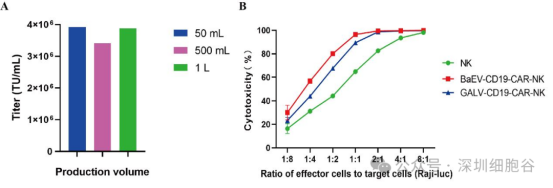
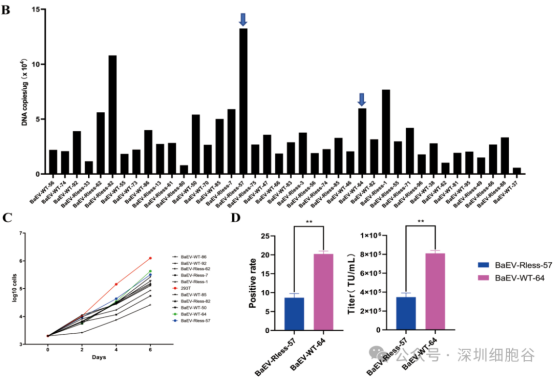
4. Scalability and Enhanced Cytotoxicity
The team further tested the titer of BaEV-CD19 CAR-RV produced under large-scale industrial conditions.
· Scalability: The system maintained high titers across production volumes of 50 mL, 500 mL, and 1 L, confirming its capability for large-scale industrial production.
· Functionality: Cytotoxicity assays revealed that CAR-NK cells prepared using BaEV-CD19 CAR-RV exhibited stronger cytotoxicity compared to those prepared using GALV-CD19 CAR-RV.
(See Figure 6: Titer and functional validation of large-scale produced BaEV-CD19 CAR-RV)
The stable cell line developed by the Shenzhen Cell Valley team enables the large-scale, stable production of BaEV-enveloped retroviral vectors. This platform supports low-cost, high-quality, and highly efficient industrial manufacturing of pseudoviruses and their application in the efficient transduction of cell therapy products.
Key Advantages over Existing Technologies:
· Vs. Transient Transfection (Lentivirus): Eliminates the need for multi-plasmid transfection, simplifying the manufacturing process, reducing costs, improving quality control, and facilitating large-scale production.
· Vs. Stable GALV Retrovirus: Achieves highly efficient transduction of hard-to-transduce primary immune cells (such as NK and T cells) at lower MOIs. This significantly increases the positive transduction rate while maintaining stability, without compromising cell viability or expansion capabilities.
This breakthrough underscores Shenzhen Cell Valley’s commitment to advancing core technologies in gene and cell therapy, paving the way for more accessible and effective treatments for patients globally.
 Scan to follow our latest news
Scan to follow our latest news Working hours: Monday to Friday, 9:00-18:00
Contact:Ms. Lai
Email:laijiaqi@sz-cell.com
Address:No. 1, Rongtian Road, Jinsha Community, Kengzi Street, Pingshan District, Shenzhen, China (Hepure Biomedical Ecological Park)