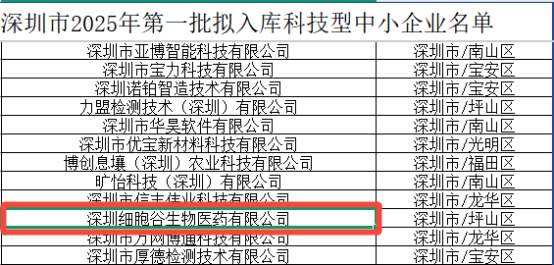
Recently, the Shenzhen Municipal Bureau of Industry and Information Technology announced the list of technology-based small and medium-sized enterprises (SMEs) selected for the first batch of 2025. Shenzhen Cell Valley has been honored with this designation for three consecutive years. Technology-based SMEs serve as a cornerstone of the national innovation system, renowned for their technology intensity, vibrant innovation, and rapid market responsiveness, acting as a core engine for industrial upgrading and high-quality economic development.
Adhering to its mission of "Cells for the Benefit of All, Genes Creating the Future," Shenzhen Cell Valley focuses on the cell and gene therapy sector. The company continues to increase R&D investment to solidify its core technological barriers, deepen industry-academia-research collaboration to build an open innovation ecosystem, and accelerate the industrialization of scientific achievements while cultivating high-end technical talent urgently needed by the industry.Leveraging the platform of a technology-based SME, Shenzhen Cell Valley will further integrate capital, policy, and international resources. It aims to build full-chain capabilities ranging from early-stage research and clinical validation to large-scale production. By doing so, the company remains committed to delivering safe, effective, and accessible therapeutic solutions to the industry, truly benefiting a broader range of patients.
 Scan to follow our latest news
Scan to follow our latest news Working hours: Monday to Friday, 9:00-18:00
Contact:Ms. Lai
Email:laijiaqi@sz-cell.com
Address:No. 1, Rongtian Road, Jinsha Community, Kengzi Street, Pingshan District, Shenzhen, China (Hepure Biomedical Ecological Park)